Aim
The primary objective is to compare the therapeutic effects of IOT-BPEMFE and OT in patients with KOA, providing evidence to inform the selection of optimal community-based exercise delivery mode. The primary hypothesis is that IOT-BPEMFE will achieve pain and functional outcomes that are noninferior to those achieved with OT.
Secondary objectives are to assess the effects of IOT-BPEMFE on daily living activities, psychological status, and functional physical performance over a 12-week post-randomization follow-up period. The secondary hypothesis is that IOT-BPEMFE will improve in daily living activities, psychological status, physical function, and quality of life that are noninferior to those achieved with OT. Furthermore, any clinically significant differences in outcomes between IOT-BPEMFE and OT will be maintained at the 12-week follow-up.
Design
This study will be a randomized, controlled, noninferiority clinical trial with blinded outcome assessment and a 12-week follow-up. The SPIRIT checklist will guide reporting [20].
Participants
The inclusion criteria will be as follows: 1. A diagnostic of KOA according to the criteria in [21]: (1) The participant must have experienced repeated knee pain in the past month; (2) X-ray images taken in a standing or weight-bearing position must show narrowing of the joint space, subchondral bone sclerosis, and/or cystic change, and the presence of osteophyte at the joint margin; (3) Age ≥ 40 years; (4) morning stiffness ≤ 30 minutes; (5) a sensation or sound of bone friction during activity. KOA is diagnosed if either both criteria (1) and (2) are met, or if all of the criteria (1), (3), (4), and (5) are satisfied; 2. Kellgren-Lawrence (K-L) grade II – III [22]; 3. The participant should demonstrate good cognitive function, compliance, and cooperation in completing various assessments and treatments; 4. The participant should not have any serious systemic diseases, and their general health should be able to tolerate exercise training; 5. The participants have the ability to independently or with assistance, operate the APP on a mobile phone; 6. The participant must volunteer for this study and sign an informed consent form.
The exclusion criteria will be as follows: 1. Severe knee joint deformity; 2. Age ≥ 75 years; 3. Cognitive impairment, significant mental health disorder, or any other condition affecting lower limb function or exercise tolerance; 4. Diagnosis of rheumatoid arthritis or suspicion of other knee joint pathologies; 5. Significant cardiovascular disease or other severe comorbidities that pose a risk to participation.
Study setting and recruitment
The study will be conducted in the outpatient physiatry and inpatient wards of the Department of Rehabilitation, the Department of Orthopedics, and the Fourth Medical Center of PLA General Hospital. A well-trained physiatrist (HYS), unaware of the participants’ allocation, will screen all potential participants before they enter the study. The general study process, along with the responsibilities of both participants and researchers, will be explained to potential participants or their guardians. Individuals who understand the study’s purposes and agree to participate will sign an informed consent form, which includes information about their right to withdraw at any time. Baseline demographic data, KOA medical history, and prior treatments will be recorded on the same day. Two blinded assessors (L. J. S. and Z. X. H.) will collect baseline measures (T0) before randomization. During the trial, these assessors will also conduct follow-up assessments at the following time points: (1) immediately following the 2-week post-randomization (midterm, T1), (2) 4-week post-randomization (post-intervention T2), and (3) 12-week post-randomization (long-term follow-up, T3). Participants will attend clinic follow-up visits at 2 weeks, 4 weeks, and 12 weeks. The CONSORT (Consolidated Standards of Reporting Trials) [23] flowchart is illustrated in Fig. 1. The recruitment period was 12 weeks.
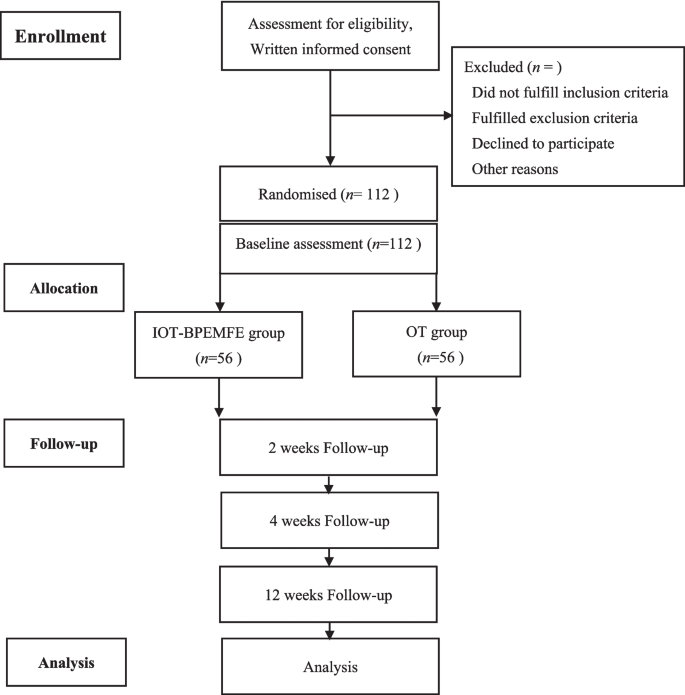
CONSORT flowchart of the study
Sample size
Allen et al [24] investigated the effects of Internet-based exercise training on patients with KOA, and the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scale was chosen as the primary evaluation index. The mean ± SD of the WOMAC score was 6.06 ± 3.66 for the intervention group and 6.73 ± 3.59 for the control group. A noninferiority test of two independent sample means was conducted, with a noninferiority margin of 3, α = 0.025, and 1-β = 0.8, by inputting the above parameters in PASS 20.0 (power analysis and sample size) software. Accounting for a 15% attrition rate, a minimum sample size of 112 participants (56 per group) was determined.
Randomization and blinding
Using a computerized random number generator, participants will be randomly assigned to the intervention or control groups in a 1:1 allocation ratio. The allocation sequence will be generated by a researcher (Y. N. X.) who is not involved in recruitment or allocation, and the results will be sealed in opaque envelopes to ensure the fairness of participant grouping. Participant recruitment will be conducted by a physician (J. Q.) unaware of the allocation sequence, and all participants will receive counseling under the researcher’s supervision. Assessors and statisticians will be blinded to group assignment. While binding of participants is not feasible due to inherent differences in intervention settings between the IOT-BPEMFE and OT groups, participants will remain unaware of the study’s hypothesized outcome (i.e., they will not be informed which group is expected to demonstrate superior efficacy).
Blinding safeguards: Assessor blinding will be maintained through standardized outpatient assessments using identical equipment/protocols, strict isolation of group labels and intervention histories, and separation of assessment/rehabilitation teams. Participants will be instructed not to disclose their intervention setting and will use identical clinic check-in procedures. All data will be protected using a multidimensional anonymization technology system to ensure privacy. During the data preprocessing stage, the system will replace the “home/clinic” identifier with neutral labels (model A/B). For physiological parameters collected by IOT devices, a key separation mechanism will be established between the device ID and the subject’s unique identifier. All data storage will adopt encrypted storage and permission-based access controls to ensure that data access is fully auditable and in compliance with hospital data management security and regulatory standards. Statisticians will analyze encrypted datasets (group X/Y).
Intervention
All participants will receive the same intervention: PEMFs therapy for 30 min twice daily and exercise for 30 min once daily, 5 days a week for 4 weeks.
Before the intervention, all participants will be informed about the study procedures and potential risks associated with PEMFs therapy for KOA and allowed to ask questions. During treatment, participants will sit or lie supine on a treatment table with the knee flexed and supported by a pillow. Under standard clinical conditions, with routine hygiene preparation and real-time parameter monitoring, the PEMFs treatment coils will be positioned around the knee.
Both groups will receive consistent PEMFs therapy using the same device model and parameter settings throughout the study. The device operates at a frequency of 10–30 Hz, with a 50%–70% duty cycle, consuming 160 VA with a 50-W peak output power, generating a therapeutic PEMFs intensity of 10–30 mT. The device, controlled via a panel with 27 or more settings allowing for various parameter combinations, uses 3 pads that can generate fields positioned directly over the treatment area to transfer energy to the tissue. In this study, the device will be set to 30-mT intensity and 15-Hz frequency for treatment. All participants in both groups will be informed that they should not experience any unusual sensations, such as pain, burning, coldness, or numbness, during the PEMFs intervention [25].
Exercise programs will be tailored to individual participants based on assessments of pain, function, and current activity levels. The programs include strengthening exercises (quadriceps, adductors, hamstrings), stretching exercises (quadriceps and gluteus medius), and aerobic activity, all in accordance with the current guidelines for physical activity set by the United States Department of Health and Human Services [26]. Participants will be encouraged to perform strength and stretching exercises five times per week and to engage in daily or frequent aerobic exercises. To prevent sports injuries, warm-up and cool-down activities are necessary. It is recommended that the duration of exercise to achieve the desired intensity is 30 min.
IoT-based pulsed electromagnetic field combined exercise group (IOT-BPEMFE)
The custom-designed “Kangfuxing (KFX)” application (app) facilitates the delivery of an IoT-based rehabilitation program combining PEMFs therapy and exercise, guided by Osteoarthritis Research Society International guidelines [7]. The app includes interfaces for both clinicians and participants, and provides instructions for participants with KOA on using the “KFX” app. Participants are instructed to install the “KFX” app immediately after randomization and to continue using it throughout the 12-week follow-up period.
The PEMFs device will be delivered directly to the patient’s home via express delivery, eliminating the need for the patient to carry it. Manufactured by Better Health Corporation (Sichuan, China), the device has received approval from the National Medical Products Administration (registration number 20162090198) for managing KOA (see Fig. 2).

The pulsed electromagnetic fields device. (A) Device application diagram, (B) operating machine and three pads, and (C) operating machine that generates fields above the therapeutic site. Figure is owned by Better Health Corporation, Sichuan, China
The exercise therapy will use videos and photographs within the “KFX” app to demonstrate proper exercise technique. Participants can initiate treatment by turning on the “KFX” app and scanning the QR code on the PEMFs device. No manual adjustments are required, and treatment parameters (such as prescription, frequency, and duration) are automatically displayed.
The “KFX” app will send daily notifications to participants. If participants fail to log in for 1 day, they will receive automated reminders to engage with the platform. If participants do not complete their exercise and PEMFs treatments on a given day, they will receive reminders the following day. If tasks remain incomplete for two consecutive days, the system will alert the physician, who will then contact the participants via phone or video call to assess their status. The physician will have a video call each week with the participant to evaluate their condition and record symptom responses. The participant’s progress will be tracked by the app, which will display pain levels, functionality, and exercise over time through graphs or text. The PEMFs device was returned via express delivery after 4 weeks of intervention (Fig. 3).
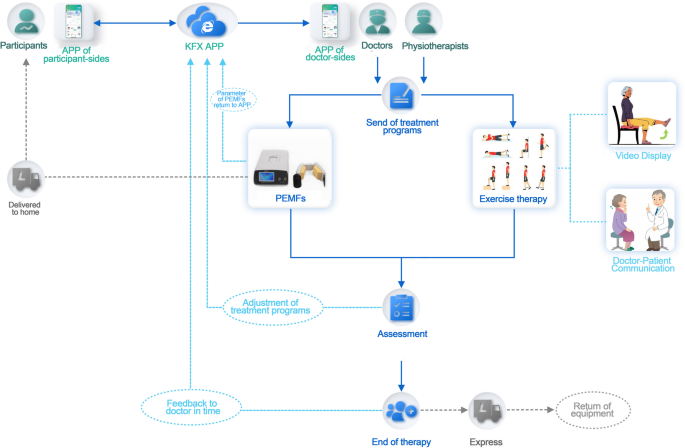
Overview of the intervention groups
Outpatient therapy group
Participants in the OT group will receive the same protocol of PEMFs therapy and exercise as the other group, but the treatments will be administered in the outpatient department.
Participants in both groups may withdraw from the study at any time; however, withdrawal rates and reasons will be recorded. If their symptoms worsen, participants may receive additional treatments, and any additional treatments received during the follow-up period will be documented.
Measures
Table 1 shows the assessment schedule. Outcome measures will be assessed at three time points post-baseline.
Definition of outcomes
The primary outcome will be knee osteoarthritis symptoms and functional status, measured using the Western Ontario and WOMAC, with total scores ranging from 0 to 96. Secondary outcomes will include pain intensity (Numerical Rating Scale, NRS, 0–10), activities of daily living (Instrumental Activity of Daily Living, IADL, 0–87), functional physical performance (Timed Up and Go, TUG, in seconds), psychological status (Self-Rating Anxiety Scale, SAS, and Self-Rating Depression Scale, SDS, both up to 80), quality of life (36-Item Short Form Health Survey, SF-36), muscle strength (manual muscle test (MMT), via dynamometer), range of motion (ROM, measured by goniometer), knee joint function (Knee Society Score, KSS, up to 100), and physical function limitations (Knee Injury and Osteoarthritis Outcome Score, KOOS, 0–68). Additionally, patient satisfaction (assessed via a department-developed questionnaire) and adverse events (documented throughout the study) will be analyzed. All outcomes will be assessed at baseline (T0), 2 weeks (T1), 4 weeks (T2), and 12 weeks (T3), with mean changes from baseline used for continuous measures and descriptive statistics for categorical data.
Primary outcome
WOMAC will be used to assess lower extremity pain (5 items), stiffness (2 items), and function (17 items). Each item is scored on a 0–4 Likert scale (0 = no symptoms; 4 = extreme symptoms), resulting in a total score range of 0–96, with higher scores indicating worse symptoms [27].
Secondary outcomes
Pain intensity: Average knee pain intensity over the last 48 h will be evaluated using a NRS, where 0 represents “no pain” and 10 represents “maximal pain.”
Activities of daily living: IADL will be assessed using a performance-based evaluation and semi-structured interview. This instrument comprises 29 items across eight domains, scored on a 0–3 scale (0 = complete independence; 3 = complete dependency) [28].
Functional physical performance: TUG test, measured in seconds, will be used; shorter times indicate better performance [29].
Psychological status: SAS [30] and SDS [31] scores will be assessed. Each scale contains 20 items, with total scores ranging up to 80. Higher scores indicate greater severity of anxiety/depressive symptoms.
Quality of life: SF-36 will be utilized as a validated health status measure consisting of 36 questions. The composite score reflects a continuum from poor to excellent quality of life [32].
MMT: Muscle strength of the quadriceps, hamstrings, iliopsoas, and gluteus medius will be assessed using a handheld dynamometer. This method demonstrates established reliability in KOA populations (intraclass correlation coefficient = 0.92 for extensor; 0.75 for flexor) [33]. Each muscle group will be tested twice, with mean values calculated for analysis.
ROM: Active and passive knee flexion/extension angles will be measured using a goniometer. Measurements will be repeated twice per motion plane, and the average will be recorded.
KSS: The KSS [34] will be applied to evaluate knee joint functional status; total scores range up to 100, with higher scores indicating better function.
KOOS: Physical functional limitations during the past week will be assessed using the KOOS [35]. Total scores range from 0 (no dysfunction) to 68 (severe dysfunction).
Adverse event monitoring: All adverse events will be meticulously documented, including their cause, treatments, outcome, and relationship to the intervention. Site researchers will report any adverse events to an independent data safety monitoring committee at the leading hospital for further evaluation and management.
Patient satisfaction: A department-developed satisfaction questionnaire will be administered exclusively to the IOT-BPEMFE group participants to evaluate satisfaction (Fig. 4).
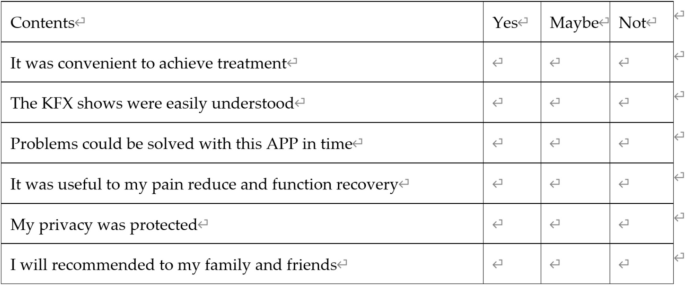
The satisfaction survey of participants in the IOT-BPEMFE group
Data collection baseline assessment
Data collection will adopt a dual-track system of structured electronic case report forms and paper documents to ensure original data integrity, traceability, and readability. Records must be entered in real time and undergo a dual-person verification mechanism, with modification traces recorded throughout the process. During the data transformation, data will undergo cleaning and standardization, and data formats will be unified and integrated into the analysis platform. Data storage will combine encrypted cloud servers with local backups, with tiered permission management and audit trials to ensure traceability.
Baseline assessments will be conducted after obtaining informed consent. Data collected will include the following: demographic characteristics (age (calculated from ID card birthdate), gender, height, weight, body mass index (BMI), education level, occupation, hobbies, marital status, smoking/alcohol habits) and clinical history (medical/surgical history, current medication regimen).
Throughout the study, we will monitor and record recruitment, attendance, and follow-up rate. Reasons for exclusions, declining participation, and dropout will be documented, along with any challenges and obstacles encountered. Adverse events and healthcare concerns will be recorded in a study log.
Plans for protocol modifications
Any major amendments to this protocol, such as those involving safety adjustments or primary endpoint modifications, will strictly adhere to ethical review procedures. The Ethics Committee on Biomedical Research of the Fourth Medical Center of PLA General Hospital, the Chinese Clinical Trial Registry (ChiCTR), and all researchers will be notified simultaneously, and the ethical review documentation will be updated and submitted to the registration platform for filing. If the amendments affect the treatment plan or risk perception of participants, they will be informed in writing and followed by signing a new informed consent document before implementation.
Plans to give access to the full protocol, participant-level data, and statistical code
The study protocol and its core findings will be published in a peer-reviewed academic journal. To protect participants’ privacy, all raw data and statistical codes will be anonymized and not publicly released with the paper. Research data management complies with ethical review requirements. After approval by the ethics committee and signing a data sharing agreement, de-identified analysis datasets and statistical codes may be obtained from the corresponding author through a formal application process.
Data analysis
A blinded data analyst will perform all analyses using the Statistical Package for Social Science, version 20.0 (SPSS Inc., Chicago, IL, USA). Prior to statistical analysis, data will be tested for normality as per the assumptions of parametric statistics. Baseline participants’ characteristics will be presented using descriptive statistics. Noninferiority will be declared if the lower bound of the two-sided 95% confidence interval (CI) for the between-group difference (IOT-BPEMFE minus OT) is −3 units for the change in WOMAC at 4-week post-randomization. The between-group differences and 95% CI for weeks 2,4 and 12 will be calculated using mixed linear models (treatment group versus time). Correlations between data on treatment effect (e.g, adherence、magnitude of pain reduction) and outcome measures(e.g., knee function scores such as WOMAC) will be analyzed using linear regression analysis in both groups.
The multiple imputation method in SPSS will be used to address missing data [26], and an intention-to-treat analysis (including all randomized participants) will be conducted. No adjustments for multiplicity will be made, and the analysis will be conducted by the statisticians in the independent data safety monitoring committee.
link

