Data source
This retrospective observational study utilized data extracted from the eICU Collaborative Research Database (eICU-CRD), a multicenter, high-granularity ICU database containing de-identified clinical records of over 200,000 adult ICU admissions across the United States between 2014 and 2015 [9].The eICU-CRD aggregates electronic health records from ICUs monitored by the Philips Healthcare eICU program, capturing comprehensive patient demographics, laboratory results, vital signs, treatment interventions, and outcomes [9, 10].
Access to the database was authorized after completion of the Collaborative Institutional Training Initiative (CITI) program’s “Data or Specimens Only Research” module and certification by the PhysioNet Review Board. Institutional review board approval from the Massachusetts Institute of Technology (our record ID: 13926093) was waived due to the study’s retrospective design, absence of direct patient intervention, and compliance with HIPAA Safe Harbor provisions, as validated by Privacert (Cambridge, MA), which certified the dataset’s de-identification standards [11]. Informed consent was exempted under these conditions. The study adhered to the Declaration of Helsinki and followed STROBE guidelines for observational research [12].
Study population
This retrospective cohort study utilized data from the eICU Collaborative Research Database (eICU-CRD). Patients aged ≥ 60 years with a diagnosis of diabetes mellitus admitted to the ICU were initially screened. Diabetes diagnosis was established using validated ICD-9-CM diagnosis codes within the database. The AIP was calculated as log10 (triglycerides/high-density lipoprotein cholesterol) using lipid measurements obtained within 24 h of ICU admission (± 24 h window).
Exclusion criteria included: (1) non-first ICU admission, (2) ICU length of stay < 48 h, (3) age < 60 years or ≥ 120 years (to exclude outliers), (4) missing 28-day mortality data, (5) unavailable TG or HDL-C measurements for AIP calculation, and (6) incomplete glycemic control records (HbA1c or fasting glucose). The final cohort comprised 7873 elderly diabetic patients (Fig. 1). All analyses adhered to the STROBE guidelines for observational studies.
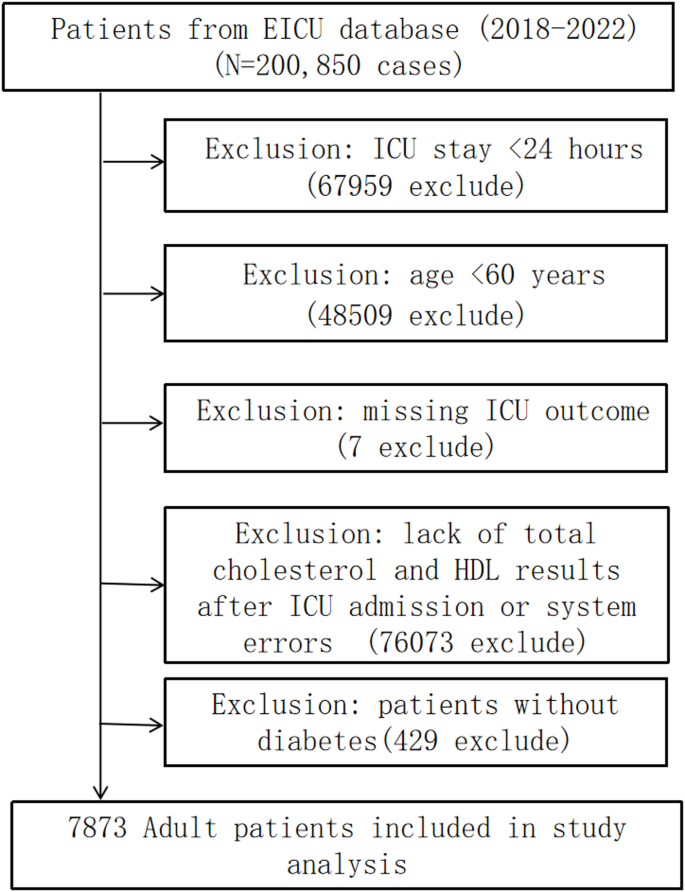
Flowchart of patient selection from the eICU collaborative research database
Variables
Baseline demographic and clinical data (age, sex, comorbidities, vital signs, laboratory measurements) were extracted. Variable selection employed hierarchical criteria:1) Clinical relevance (age, sex, ethnicity); 2) Data integrity and literature support (comorbidities including COPD [13, 14], pneumonia [15], arrhythmia [16, 17], CHF [18], and AMI [19, 20], exhibiting < 1% missingness and established associations with ICU mortality in published literature); 3) Statistical feasibility (condition prevalence > 5%, variance inflation factor [VIF] < 2.5 confirming minimal collinearity) (Table S1, Table S2). The primary outcome was 28-day mortality.
Statistical analysis
Descriptive data analysis
Continuous variables were expressed as median (interquartile range) or mean ± standard deviation based on the Shapiro-Wilk normality test. Categorical variables were reported as frequencies (percentages). Inter-group differences across AIP tertiles were analyzed using one-way ANOVA or the Kruskal-Wallis test for continuous variables; chi-squared test was used for categorical variables.
Association model construction
Generalized additive models (GAM) with cubic spline functions were employed to evaluate non-linear relationships between AIP and 28-day mortality. Subsequently, multivariable logistic regression quantified this association, generating adjusted odds ratios (ORs) with corresponding 95% confidence intervals (95% CIs).
Threshold effect analysis
Piecewise linear regression modeling identified mortality risk threshold effects. The inflection point was determined via maximum likelihood estimation, implementing grid search across the predefined AIP interval [-0.6, 2.0] [13]. Model adequacy was verified by likelihood ratio testing comparing segmented and linear specifications. Bootstrap resampling (1,000 iterations) derived 95% CIs for the inflection point.
Sensitivity analysis
Robustness was evaluated through two complementary approaches: multiple imputation using random forests for variables with < 5% missing data, followed by comprehensive model re-estimation; subgroup analysis excluding cirrhotic patients (n = 837; ICD-9 codes). We assessed the potential for unmeasured confounding in the association between AIP and 28-day mortality by calculating E-values [14]. E-values quantified the minimum unmeasured confounder strength needed to explain the observed AIP-mortality association, accounting for covariates [15, 16]. All analyses were conducted in EmpowerStats v5.0 and R v3.6.1 (packages: mgcv v1.8-42, segmented v1.6-4) with statistical significance defined at α = 0.05 (two-tailed).
link

